|
This activity should follow a full lesson on Lewis Structures, including exceptions to the octet rule.However, the visual focus of this activity does seem to help many of my students grasp the concept of shapes of molecules. It may need to be slightly modified for lower level classes, especially if Lewis structures are difficult for them. I have used this lesson with AP Chemistry classes, college-prep Chemistry II, and college-prep Chemistry I.If you are using latex balloons, check to see if any students have latex allergies/sensitivities.String or rubber bands (can be helpful for tying balloons together).Valence Shell Electron Pair Repulsion Theory.This activity supports students’ understanding of Organize their information in the form of a chart.Explain how the geometry of a molecule helps to determine its properties.State the two factors that determine the polarity of a molecule.Show how molecular geometry is a function of electron pair geometry.Use balloons to illustrate the different electron pair geometries around the central atom.Name and describe the five electron pair geometries which can surround the central atom.Differentiate electron pair geometry from molecular geometry.Define the VSEPR theory and explain its relationship to the shape of molecules.Explain electron properties of moleculesīy the end of this activity, students should be able to.Explain structural properties of molecules.SAP-4.C: Based on the relationship between Lewis diagrams, VSEPR theory, bond orders, and bond polarities:.Topic 2.7: VSEPR and Bond Hybridization.SAP-4.B: Represent a molecule with a Lewis diagram that accounts for resonance between equivalent structures or that uses formal charge to select between nonequivalent structures.SAP-4.A: Represent a molecule with a Lewis diagram.SAP-3.B: Represent the relationship between potential energy and distance between atoms, based on factors that influence the interaction strength.Topic 2.2: Intramolecular Force and Potential Energy.SAP-3.A: Explain the relationship between the type of bonding and the properties of the elements participating in the bond. 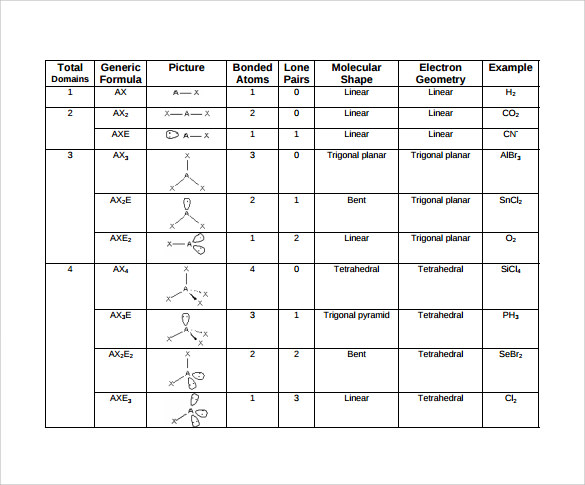
Unit 2: Molecular and Ionic Compound Structure and Properties.SAP-1.A: Represent the electron configuration of an element or ions of an element using the Aufbau principle. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
